Epidiolex: A Milestone in Cannabis-Derived Medication
Introduction
In a landmark decision that underscores the evolving landscape of cannabis research and its medical applications, the U.S. Food and Drug Administration (FDA) has approved Epidiolex, a medication derived from cannabis. This approval marks a significant milestone in the recognition of cannabis’s potential in the medical field. Epidiolex, specifically designed to treat rare and severe forms of epilepsy, is a beacon of hope for patients and a testament to the therapeutic possibilities of cannabis-derived medications.
The Journey of Epidiolex
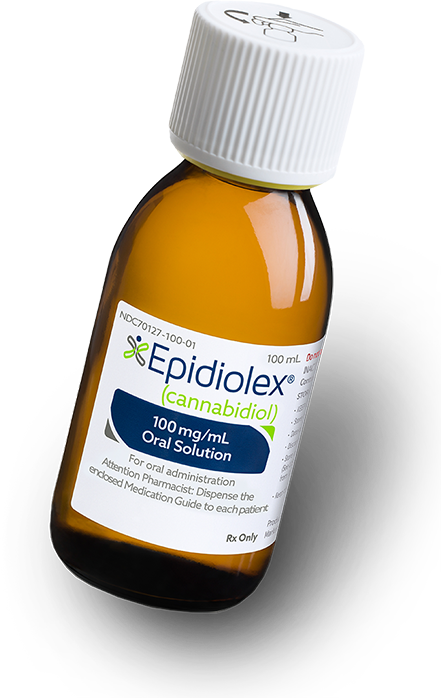
Epidiolex, an oral solution containing cannabidiol (CBD), is the first FDA-approved drug that contains a purified drug substance derived from marijuana. It is designed to treat seizures associated with two rare and severe forms of epilepsy: Lennox-Gastaut syndrome and Dravet syndrome, in patients two years of age and older.
Understanding the Medical Breakthrough
CBD, a chemical component of the Cannabis sativa plant, is known for its therapeutic properties without the intoxicating effects typically associated with marijuana. Unlike tetrahydrocannabinol (THC), CBD does not cause the euphoria or “high” but has been found effective in reducing the frequency of seizures.
Clinical Trials and Approvals
Epidiolex’s effectiveness was demonstrated in three randomized, double-blind, placebo-controlled clinical trials involving 516 patients. These trials showed that Epidiolex, when taken with other medications, effectively reduced the frequency of seizures compared to a placebo. The most common side effects included sleepiness, sedation, lethargy, elevated liver enzymes, and decreased appetite.
Epidiolex: A New Hope for Patients
The approval of Epidiolex is particularly significant for patients with Dravet syndrome and Lennox-Gastaut syndrome. These conditions, often appearing in the first few years of life, lead to multiple types of seizures and can have a profound impact on the quality of life.
Addressing Rare Epileptic Conditions
Dravet syndrome, a rare genetic condition, and Lennox-Gastaut syndrome, characterized by multiple seizure types, have been challenging to treat. Epidiolex offers a new, FDA-approved treatment option, providing a significant improvement in the therapeutic approach to these complex conditions.
For families and patients dealing with these severe forms of epilepsy, Epidiolex represents more than just a medication; it’s a ray of hope. It offers a chance for better control of seizures, improved quality of life, and a step towards normalcy in their daily lives.
Regulatory and Legal Aspects
The approval of Epidiolex also highlights the evolving regulatory landscape regarding cannabis-derived medications. Under the Controlled Substances Act (CSA), CBD is a Schedule I substance due to its origin from the cannabis plant. However, the FDA’s approval and the clinical evidence supporting Epidiolex’s efficacy could influence future regulatory decisions.
The FDA’s approval of Epidiolex is a reminder of the importance of rigorous scientific research and drug development in the field of cannabis-derived medications. It opens the door for further research and development in this area, potentially leading to more treatments derived from cannabis.
Conclusion
The approval of Epidiolex marks a historic moment in the recognition and acceptance of cannabis-derived medications. It not only offers new treatment options for those suffering from rare and severe forms of epilepsy but also paves the way for future research into the medical applications of cannabis. As we witness this groundbreaking development, we invite our readers to share their thoughts and experiences. How do you think Epidiolex will impact the future of medical treatment? What are your views on the use of cannabis-derived medications? Join the conversation in the comments below.

